12-section investment thesis
346 paragraphs of analyst-grade narrative: strategic context, market structure, demand drivers, technical feasibility, competitive landscape, federal funding stack, capital stack and unit economics, key personnel, regulatory pathway, comparable transactions, risk factors, and investment recommendation. Four appendices.
10-sheet primary-source fact sheet
Structured xlsx with every data point cited in the thesis: USGS-equivalent FDA Drug Master File records, supply chain concentration, market size and pricing, policy timeline, federal funding stack, competitive landscape, end-use demand, regulatory, sources (25 numbered citations), and modeling assumptions.
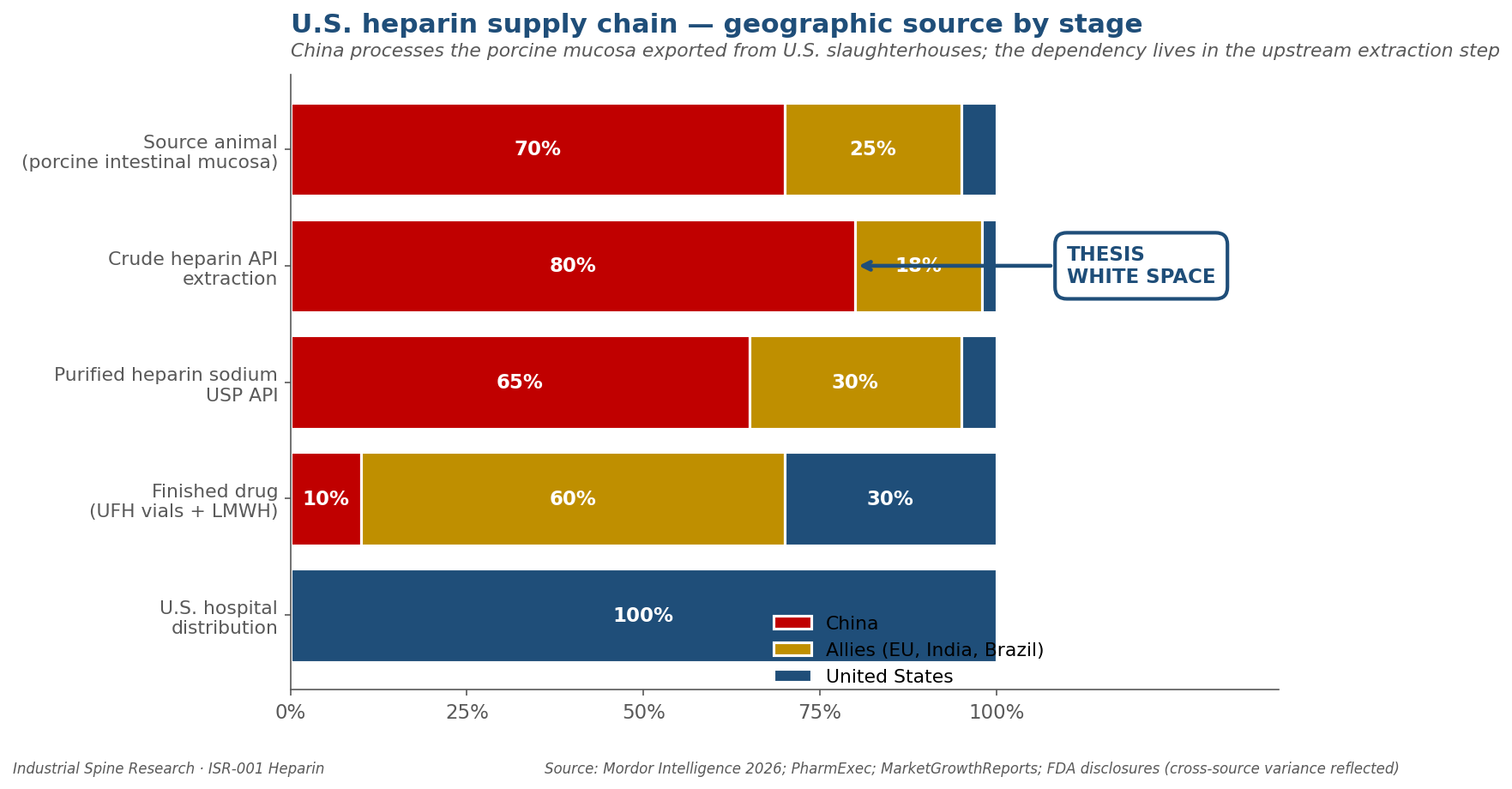
Three production charts
Print-ready 300 DPI PNG charts: U.S. heparin supply chain geographic source by stage, global market forecast convergence across five analyst sources, and 18-year policy/risk chronology from BSE bovine discontinuation to BARDA BioMaP solicitation.